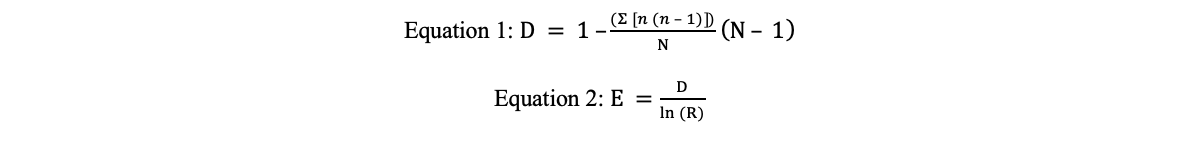- Home
- About Me
- Theme 1
- Theme 2
- Theme 3
-
Theme 4
-
Theme 5
- Application of Bloom’s Taxonomy in Outdoor Learning School
- Assessment Informing Instruction
- Developmental Pedagogy Assessment (LDPA) Question 5
- Reflections >
- An Evaluation of Systems Thinking in the 2021 TREE Semester Curriculum
- Recommendations for Equity-Focused Environmental Education Program Evaluations
The proposed Anthropocene Epoch (Lewis & Maslin 2015) results from human impact on the Earth. Scientists argue that humanity is currently the largest contributing factor to the fundamental alteration of earth systems, including lakes (Zalasiewicz et al., 2021). The proximity of high alpine shallow lakes to headwaters makes them important indicators of human impacts on the climate. Understanding shallow lake dynamics provides background information that is necessary to make decisions about managing shallow lakes and maintaining lake health. Climate change is causing many high alpine regions to become hotter and dryer. These changes result in shallow lakes losing ice cover, increasing evaporation and water loss, increasing water temperatures, and increasing changes to mixing regimes. Changes in water budgets (availability of water in a lake’s watershed) are influenced by many environmental factors such as precipitation, evaporation, and changes in terrestrial water storage (runoff and snow/soil water storage). As the climate warms and dries, evaporation in lakes is projected to increase (Woolway, 2020). Increasing levels of evaporation and decreasing water budgets are likely to result in increased lake succession. Thus, climate change will likely speed up the transition from shallow lakes to wetlands.
Biodiversity
Human activities have significant effects on global biodiversity. Reducing biodiversity of aquatic systems often reduces the system’s ability to be resilient in the face of environmental change (e.g., Pelletier et al. 2020; Penaluna et al. 2017). Climate change, introduction of exotic species, land use, and changes in atmospheric CO2 and/or nitrogen deposition are the major causes of projected changes in biodiversity for biomes (Chapin et al., 2000). It is not unreasonable to assume that these causes could also impact biodiversity in shallow lakes, specifically on macroinvertebrate populations. The influence of each cause depends on how we assume the causes interact. If the causes are assumed to interact multiplicatively, climate change is projected to be the most influential cause of biodiversity change (Chapin et al., 2000).
Bi-stability
Shallow lakes differ from their deeper counterparts in their distribution of aquatic vegetation and sediment to water interactions. These lakes are characterized through their bi-stability which is their ability to stay stable in either a clear vegetated state or a eutrophic state. Lakes in the clear state typically have low nutrient concentrations and submerged vegetation. Alternatively, eutrophic lakes have minimal vegetation, high nutrient concentrations, and significant algae blooms. Lakes in the eutrophic state have high turbidity due to algae blooms and wind disturbing sediments (Sheffer, 2009). Both states are stable, and it usually requires a large disturbance to push a lake from one state to the other. Though lakes in the clear vegetated state are often preferred over eutrophic lakes for their aesthetic, ecological, and recreational value, it is notoriously challenging to manage a eutrophic lake to become a clear vegetated lake.
Lake Structure
Horizontal Structure
Shallow lakes are divided into two zones due to variations in light penetration. The littoral zone describes the area near the edge of the lake where light can reach the lake floor. The outermost edge of the littoral zone, the zone of emergent vegetation, is dominated by various grasses that utilize resources from both the lake and atmosphere. The unvegetated zone towards the middle of the lake is the pelagic zone (McCulloch n.d.).
Vertical Structure
The Pelagic zone is divided into two subzones also due to variations in light penetration. The upper part of the pelagic zone that receives sufficient sunlight is called the trophogenic zone. Under the trophogenic zone, where respiration is greater than productivity, is the profundal zone. Where these two zones meet is the compensation point, where productivity and respiration are equivalent (McCulloch n.d.).
Three experiments were performed to review the current health of two shallow lakes located at the Catamount Center’s Catamount Mountain Campus outside of Woodland Park, Colorado. The goals of the experiments were to identify the current state (clear vegetated or eutrophic) of each lake and consider appropriate management strategies. One strategy for slowing succession, managing, and increasing the recreational value of the lakes is to remove the vegetation, that can impede recreational uses like boating and swimming in the littoral zones of both lakes. This paper hypothesizes that the removal of plants from the zone of emergent vegetation in shallow lakes will likely reduce the speed of succession while potentially sending the lake into a eutrophic state.
Biodiversity
Human activities have significant effects on global biodiversity. Reducing biodiversity of aquatic systems often reduces the system’s ability to be resilient in the face of environmental change (e.g., Pelletier et al. 2020; Penaluna et al. 2017). Climate change, introduction of exotic species, land use, and changes in atmospheric CO2 and/or nitrogen deposition are the major causes of projected changes in biodiversity for biomes (Chapin et al., 2000). It is not unreasonable to assume that these causes could also impact biodiversity in shallow lakes, specifically on macroinvertebrate populations. The influence of each cause depends on how we assume the causes interact. If the causes are assumed to interact multiplicatively, climate change is projected to be the most influential cause of biodiversity change (Chapin et al., 2000).
Bi-stability
Shallow lakes differ from their deeper counterparts in their distribution of aquatic vegetation and sediment to water interactions. These lakes are characterized through their bi-stability which is their ability to stay stable in either a clear vegetated state or a eutrophic state. Lakes in the clear state typically have low nutrient concentrations and submerged vegetation. Alternatively, eutrophic lakes have minimal vegetation, high nutrient concentrations, and significant algae blooms. Lakes in the eutrophic state have high turbidity due to algae blooms and wind disturbing sediments (Sheffer, 2009). Both states are stable, and it usually requires a large disturbance to push a lake from one state to the other. Though lakes in the clear vegetated state are often preferred over eutrophic lakes for their aesthetic, ecological, and recreational value, it is notoriously challenging to manage a eutrophic lake to become a clear vegetated lake.
Lake Structure
Horizontal Structure
Shallow lakes are divided into two zones due to variations in light penetration. The littoral zone describes the area near the edge of the lake where light can reach the lake floor. The outermost edge of the littoral zone, the zone of emergent vegetation, is dominated by various grasses that utilize resources from both the lake and atmosphere. The unvegetated zone towards the middle of the lake is the pelagic zone (McCulloch n.d.).
Vertical Structure
The Pelagic zone is divided into two subzones also due to variations in light penetration. The upper part of the pelagic zone that receives sufficient sunlight is called the trophogenic zone. Under the trophogenic zone, where respiration is greater than productivity, is the profundal zone. Where these two zones meet is the compensation point, where productivity and respiration are equivalent (McCulloch n.d.).
Three experiments were performed to review the current health of two shallow lakes located at the Catamount Center’s Catamount Mountain Campus outside of Woodland Park, Colorado. The goals of the experiments were to identify the current state (clear vegetated or eutrophic) of each lake and consider appropriate management strategies. One strategy for slowing succession, managing, and increasing the recreational value of the lakes is to remove the vegetation, that can impede recreational uses like boating and swimming in the littoral zones of both lakes. This paper hypothesizes that the removal of plants from the zone of emergent vegetation in shallow lakes will likely reduce the speed of succession while potentially sending the lake into a eutrophic state.
Methods
Site Description
The Catamount Mountain Campus, the field site managed by the Catamount Center, is located six miles southwest of Woodland Park, Colorado, and approximately 9600 feet elevation contained “Upper” and “Lower” lakes. Both lakes are man-made. The upper lake is intermittently stream fed from first order Crystola Creek. The lower lake is fed primarily through a spillway and spring runoff. There is a dam and a small spillway located at the north end of the lower lake. The upper lake is 4 m above and 40 m south of the lower lake. The upper lake is about 3.2 acres and 9.8 ft. (3 m) at its deepest point. The lower lake is around 1.3 acres and 13 ft. (4 m) at its deepest point. The lakes are used primarily for recreation and education (Kochevar, 1987).
Field Sampling
Macroinvertebrate Collection
Lake water was sampled from the littoral zones of the upper and lower lakes at the Catamount Mountain Campus for macroinvertebrates in four locations at each lake. Both lakes were divided into four quadrants and one location was selected from each quadrant. When taking individual samples, lake water was collected in plastic bins measuring 0.5ft. x 1ft. x 1ft. at two feet from the lake edge. Each researcher reached two feet out in front of them, scraped one centimeter of the top sediment and six inches across and the lake bottom. Each bin was filled with water within two inches of full capacity. Individual macroinvertebrate organisms were sorted, identified, counted, and recorded by species using a dichotomous key.
Temperature and DO
Researchers collected measurements at the deepest part of each lake by referencing the bathymetric profile of Kochevar (1987). At each lake, temperature and dissolved oxygen (DO) data were collected with a YSI ProODO meter at one-foot intervals in each lake. A Secchi disk was used to determine the turbidity at one-foot intervals in the deepest part of the lake.
Nutrients
The two Catamount Lakes were each divided into six data collection sites. Three of the sites were in the pelagic zone and the other three sites were in the littoral zone about one meter offshore. At each site, eight dissolved oxygen bottles were rinsed with and then filled with lake water. A YSI ProODO DO meter was used to take both temperature and DO measurements in triplicate at each sampling site. In the pelagic zones, measurements were recorded about one meter under the lake surface. Of the eight bottles collected at each site, four were wrapped in aluminum foil and designated as the dark bottles. The other four were left uncovered and available to light. Solutions of 100mM NH4+ and 10mM PO43- were added to selected bottles for nitrogen and phosphorus fertilization. The first two bottles, one light one dark, were designated the control bottles and contained only lake water. Two bottles, one light one dark, had 1.0 mL of 100mM NH4+ nitrogen solution added. Two more bottles, one light one dark, had 1.0 mL of 10mM PO43- phosphorus solution added. The final two bottles, one light one dark, had 1mL each of nitrogen and phosphorus added. All the bottles, a total of 96, were left incubating in the Lower Lake for five days. A YSI ProODO meter was used to measure the baseline temperature and DO concentration of the upper and lower lakes.
Lab Analysis
After five days of incubation, a YSI BOD probe connected to a YSI Professional Plus multiparameter water quality meter was used to measure the DO content of each bottle.
Data Analysis
Calculations
Macroinvertebrates. Calculations were done according to Equations 1-2.
In Equation 1, Simpson’s diversity index (D) was calculated using n, the number of organisms counted for each species, and N, the total number of organisms counted for all species.
Species richness (R), the number of species counted, was used to calculate species evenness (E) by Equation 2.
The Catamount Mountain Campus, the field site managed by the Catamount Center, is located six miles southwest of Woodland Park, Colorado, and approximately 9600 feet elevation contained “Upper” and “Lower” lakes. Both lakes are man-made. The upper lake is intermittently stream fed from first order Crystola Creek. The lower lake is fed primarily through a spillway and spring runoff. There is a dam and a small spillway located at the north end of the lower lake. The upper lake is 4 m above and 40 m south of the lower lake. The upper lake is about 3.2 acres and 9.8 ft. (3 m) at its deepest point. The lower lake is around 1.3 acres and 13 ft. (4 m) at its deepest point. The lakes are used primarily for recreation and education (Kochevar, 1987).
Field Sampling
Macroinvertebrate Collection
Lake water was sampled from the littoral zones of the upper and lower lakes at the Catamount Mountain Campus for macroinvertebrates in four locations at each lake. Both lakes were divided into four quadrants and one location was selected from each quadrant. When taking individual samples, lake water was collected in plastic bins measuring 0.5ft. x 1ft. x 1ft. at two feet from the lake edge. Each researcher reached two feet out in front of them, scraped one centimeter of the top sediment and six inches across and the lake bottom. Each bin was filled with water within two inches of full capacity. Individual macroinvertebrate organisms were sorted, identified, counted, and recorded by species using a dichotomous key.
Temperature and DO
Researchers collected measurements at the deepest part of each lake by referencing the bathymetric profile of Kochevar (1987). At each lake, temperature and dissolved oxygen (DO) data were collected with a YSI ProODO meter at one-foot intervals in each lake. A Secchi disk was used to determine the turbidity at one-foot intervals in the deepest part of the lake.
Nutrients
The two Catamount Lakes were each divided into six data collection sites. Three of the sites were in the pelagic zone and the other three sites were in the littoral zone about one meter offshore. At each site, eight dissolved oxygen bottles were rinsed with and then filled with lake water. A YSI ProODO DO meter was used to take both temperature and DO measurements in triplicate at each sampling site. In the pelagic zones, measurements were recorded about one meter under the lake surface. Of the eight bottles collected at each site, four were wrapped in aluminum foil and designated as the dark bottles. The other four were left uncovered and available to light. Solutions of 100mM NH4+ and 10mM PO43- were added to selected bottles for nitrogen and phosphorus fertilization. The first two bottles, one light one dark, were designated the control bottles and contained only lake water. Two bottles, one light one dark, had 1.0 mL of 100mM NH4+ nitrogen solution added. Two more bottles, one light one dark, had 1.0 mL of 10mM PO43- phosphorus solution added. The final two bottles, one light one dark, had 1mL each of nitrogen and phosphorus added. All the bottles, a total of 96, were left incubating in the Lower Lake for five days. A YSI ProODO meter was used to measure the baseline temperature and DO concentration of the upper and lower lakes.
Lab Analysis
After five days of incubation, a YSI BOD probe connected to a YSI Professional Plus multiparameter water quality meter was used to measure the DO content of each bottle.
Data Analysis
Calculations
Macroinvertebrates. Calculations were done according to Equations 1-2.
In Equation 1, Simpson’s diversity index (D) was calculated using n, the number of organisms counted for each species, and N, the total number of organisms counted for all species.
Species richness (R), the number of species counted, was used to calculate species evenness (E) by Equation 2.
Nutrients. Calculations were done according to Equations 3-5.
In Equation 3, the Ra value represents autotrophic respiration. Background DO was determined by taking averages of the DO measurements at the three sample sites for each zone. The difference in oxygen levels was multiplied by one half to approximate the autotrophic respiration.
In equation 4, NPP represents the net primary productivity.
To calculate the gross primary productivity (GPP), the NPP and Ra values were added (Equation 5). The Ra, NPP, and GPP values were calculated for each pelagic and littoral zones of both the upper and lower Catamount lakes. All values were measured in mg O2 /L. The units were then converted to gC/m3/day.
In Equation 3, the Ra value represents autotrophic respiration. Background DO was determined by taking averages of the DO measurements at the three sample sites for each zone. The difference in oxygen levels was multiplied by one half to approximate the autotrophic respiration.
In equation 4, NPP represents the net primary productivity.
To calculate the gross primary productivity (GPP), the NPP and Ra values were added (Equation 5). The Ra, NPP, and GPP values were calculated for each pelagic and littoral zones of both the upper and lower Catamount lakes. All values were measured in mg O2 /L. The units were then converted to gC/m3/day.
Statistical Analysis
Data for Ra, NPP, and GPP, organized by lake, zone, and nutrients were imported from an Excel spreadsheet to IBM SPSS v. 26.0. A two-way analysis of variance (ANOVA) was used to determine the effects of lake, zone, and nutrients on the Ra, NPP, and GPP. A post-hoc Bonferroni test was used to determine significant (p < 0.05) differences among nutrients.
Data for Ra, NPP, and GPP, organized by lake, zone, and nutrients were imported from an Excel spreadsheet to IBM SPSS v. 26.0. A two-way analysis of variance (ANOVA) was used to determine the effects of lake, zone, and nutrients on the Ra, NPP, and GPP. A post-hoc Bonferroni test was used to determine significant (p < 0.05) differences among nutrients.
Results
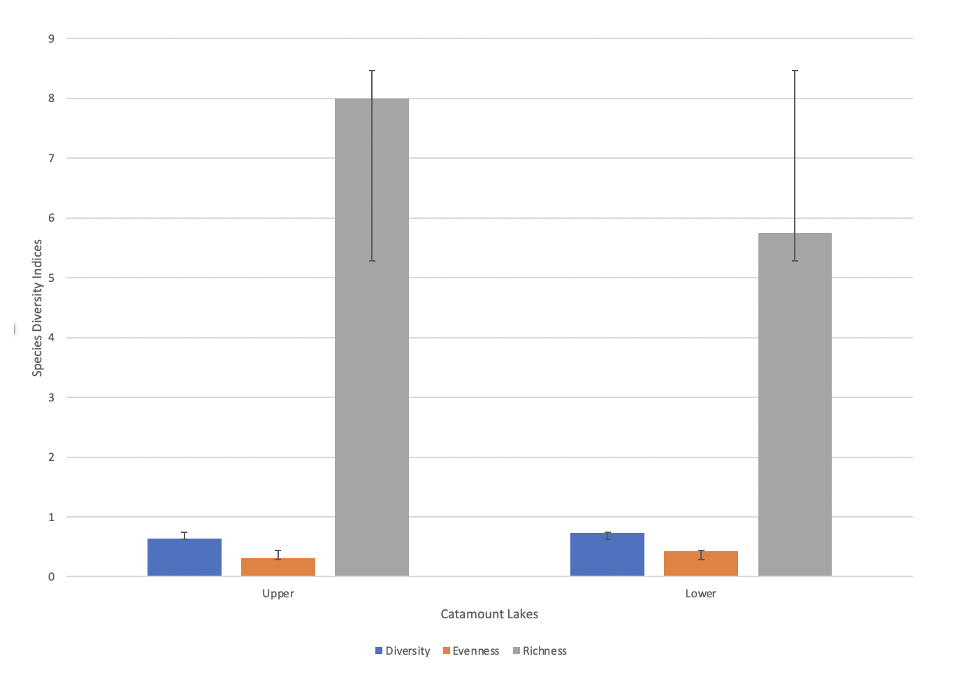
The results of all three field experiments are shown in Figures 1-4. Figure 1 depicts species evenness, richness, and diversity for both the upper and lower Catamount Lakes. The Upper Catamount Lake had visibly higher species richness, and the lower lake had more species diversity and evenness. The overlap of error bars depicts that there is no statistical difference for any of the three indices. Despite the lack of statistical differences, the two lakes are not the same. The upper lake was reported to be higher in species richness and slightly lower in both diversity and evenness.
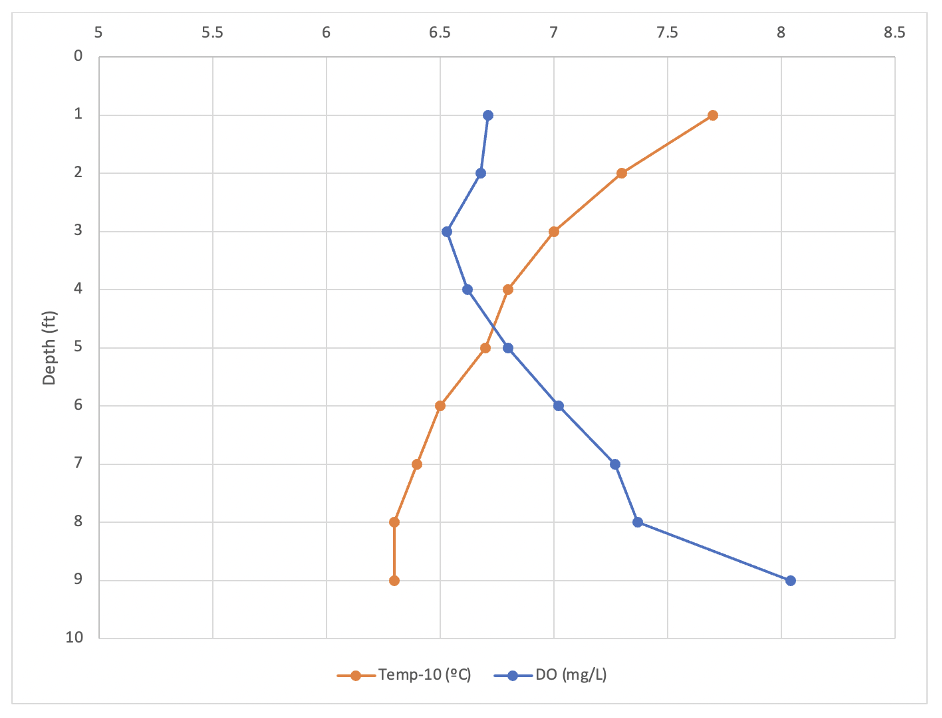
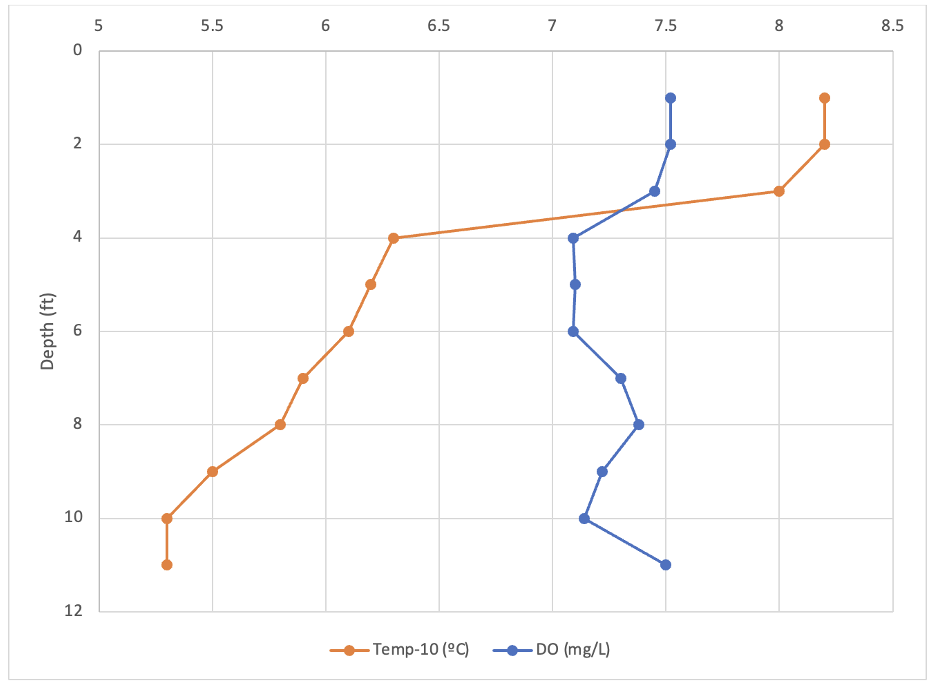
Figure 2 shows how temperature and DO change with lake depth in the upper lake. Figure 3 displays the temperature and DO for the lower lake. All temperatures displayed on the graphs were subtracted by 10 ºC to more easily compare scales. In both lakes, the temperature decreased as depth increased. In the lower lake, the temperature dropped the most from three to four feet. The upper lake surface water temperature was 17.7ºC and the deepest point was recorded at 16.3ºC. In the lower lake the surface temperature was 18.2ºC and at the bottom was 15.3ºC. The DO of the upper lake generally increased from 6.71 mg/L to 8.04 mg/L, with the lowest recorded DO of 6.53 mg/L at 3 feet deep. The DO of the lower lake showed a less clear trend, DO at the surface was 7.52 mg/L and at the bottom was 7.5 mg/L. The lowest DO measurement of the lower lake was 7.09 mg/L at four and six feet. In both lakes, the black and white of a Secchi disk was visible at all depths.
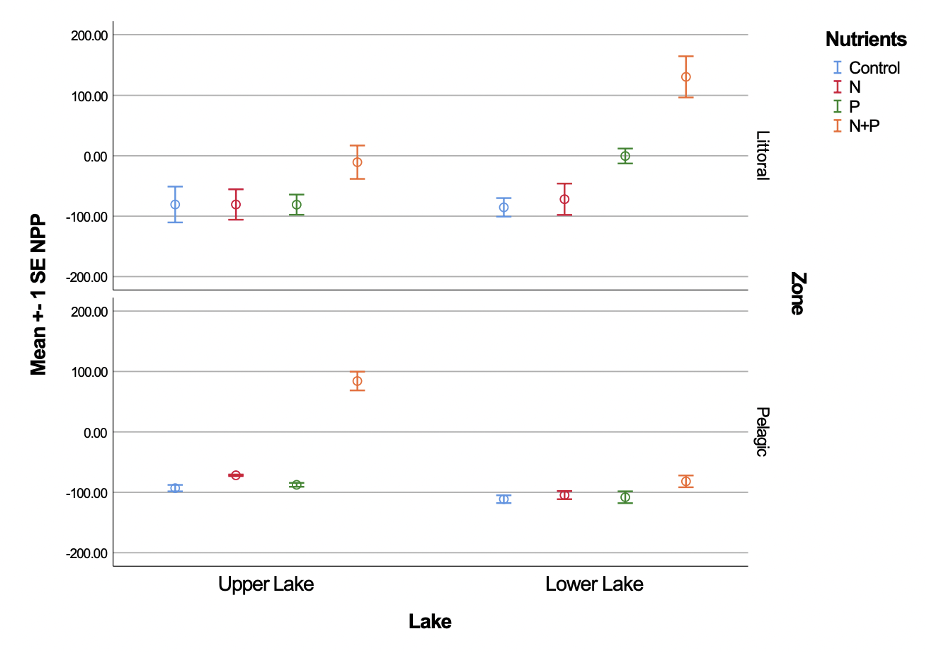
Figure 4 displays a difference in N+P from the other nutrient and control bottles. In the Upper Lake pelagic zone, the control, N, and P are all negative, while the N+P is significantly higher and positive. The greatest differences for NPP in nutrients was between the control and N+P for the Upper Lake pelagic and Lower Lake littoral zones. The least difference in NPP for the control and N+P was in the Lower Lake pelagic and Upper Lake littoral zones. The values in the littoral zone were more variable; this is displayed through larger error bars. The null hypothesis that there was no significant difference in NPP between the control and N+P was rejected (p<0.001).
Figure 2 shows how temperature and DO change with lake depth in the upper lake. Figure 3 displays the temperature and DO for the lower lake. All temperatures displayed on the graphs were subtracted by 10 ºC to more easily compare scales. In both lakes, the temperature decreased as depth increased. In the lower lake, the temperature dropped the most from three to four feet. The upper lake surface water temperature was 17.7ºC and the deepest point was recorded at 16.3ºC. In the lower lake the surface temperature was 18.2ºC and at the bottom was 15.3ºC. The DO of the upper lake generally increased from 6.71 mg/L to 8.04 mg/L, with the lowest recorded DO of 6.53 mg/L at 3 feet deep. The DO of the lower lake showed a less clear trend, DO at the surface was 7.52 mg/L and at the bottom was 7.5 mg/L. The lowest DO measurement of the lower lake was 7.09 mg/L at four and six feet. In both lakes, the black and white of a Secchi disk was visible at all depths.
Figure 4 displays a difference in N+P from the other nutrient and control bottles. In the Upper Lake pelagic zone, the control, N, and P are all negative, while the N+P is significantly higher and positive. The greatest differences for NPP in nutrients was between the control and N+P for the Upper Lake pelagic and Lower Lake littoral zones. The least difference in NPP for the control and N+P was in the Lower Lake pelagic and Upper Lake littoral zones. The values in the littoral zone were more variable; this is displayed through larger error bars. The null hypothesis that there was no significant difference in NPP between the control and N+P was rejected (p<0.001).
Figure 1: Species diversity, species richness, species evenness in the upper and lower Catamount lakes. Error bars indicate standard deviation.
Figure 2. Temperature (-10 ºC) and dissolved oxygen concentration (mg/L) as a function of lake depth for Upper Lake (Silva) on September 8, 2021.
Figure 3. Temperature (-10 ºC) and dissolved oxygen concentration (mg/L) as a function of lake depth for the Lower Lake (Risa) on September 8, 2021.
Figure 4. Net primary productivity (NPP) for control, nitrogen, phosphorus, and nitrogen + phosphorus for the littoral and pelagic zones in the upper and lower Catamount lakes. Error bars indicate standard error of the mean.
Discussion
The potential effects of removing weeds in the zone of emergent vegetation from both Catamount lakes to reduce lake succession is best evaluated through the impact on macroinvertebrate populations, turbidity, temperature/dissolved oxygen profiles, and nutrients. The data from all three experiments indicate that the two Catamount lakes are currently in clear vegetated states. The results of the Secchi disk express that there is very low turbidity in both lakes. Another indicator of a clear lake is macroinvertebrate diversity. A diversity index of 1.0 indicated infinite diversity while 0.0 indicates no diversity. The diversity index for both lakes was above 0.5 (upper: 0.64 and lower: 0.73) suggesting a diverse population of macroinvertebrates in both lakes. The nutrient data also provided evidence that the lakes are in a clear vegetated state. Eutrophic lakes tend to have higher nutrient concentrations. Our experiments on the Catamount lakes indicated that both the upper and lower lakes were co-limited by nitrogen and phosphorus. Since shallow lakes should be considered a full system, ignoring any of these factors would provide an incomplete picture of lake response to management strategies.
Periphyton in the zone of emergent vegetation uses CO2 from the atmosphere and nutrients from the lake system. If these plants are removed, nutrient availability in the lake may initially increase. Higher nutrient availability could benefit both macrophytes and algae populations. The significant difference between NPP in the N+P and control bottles provides clues about the existing nutrients in the lakes. Photosynthesis of autotrophs in the lakes can be derived from the net primary productivity. NPP was much higher in the bottles where both nitrogen and phosphorus were added. This indicates that autotrophic photosynthesis was likely limited by one or both nutrients. Figure 2 shows that NPP in the N+P was higher than either the N or P bottles. This supports the hypothesis that the lakes are co-limited by both nitrogen and phosphorus. Thus, removing the weeds as nutrient competitors may benefit other macrophytes populations in the lake. Algae are also nutrient-dependent and could benefit from less competition by periphyton for nutrients. If algae blooms are encouraged, the lake will become more turbid. Higher turbidity can prevent vegetation from reestablishing by limiting light availability in the littoral zones.
Increasing algae blooms, which increases turbidity, may also impact the temperature and DO profiles of the lakes. High turbidity results in low light penetration. If light is unable to reach below surface waters, the water underneath will not be as warm. This will result in overall colder, more stratified lakes with more extreme temperature profiles. If lakes are generally lower in temperature, there may be less evaporation of water from the lake surface. This could slow the rate of lake succession. If the algae populations increase, there will also be an increase of dying algal material that falls to the bottom of the lake. The detritivores living at the bottom of the lakes process the retired algae on the lake floor through respiration, converting food (dead algae) and DO into H2O and CO2. Increased dead algae at the bottom of the lake will increase decomposer populations and thus respiration. An increase in detritivore respiration will ultimately reduce the amount of DO, potentially resulting in an anoxic lake.
Macroinvertebrate populations may be most influenced by reductions in DO and vegetation. Lower levels of DO may ultimately inhibit macroinvertebrate populations since their respiration requires DO in the water. If other heterotrophs, such as detritivores at the lake bottom, outcompete macroinvertebrates for DO, the macroinvertebrate population may decrease. This would occur because less DO would limit macroinvertebrate respiration. It is likely that algae blooms will advantage detritivores and macroinvertebrates may die off from lack of DO in the lake. Vegetation may provide protection for macroinvertebrates from predation and disturbances. Periphyton can act as a physical barrier for macroinvertebrates, advantaging them in their predator-prey relationship with fish. This is only relevant in lakes that support fish that eat macroinvertebrates. Both the upper and lower lakes have been previously stocked with fish. All the fish in the upper lake are assumed to be dead, but the lower lake still contains some fish. Thus, this factor still may be relevant. The vegetation may also provide protection to macroinvertebrates from abiotic disturbances like wind.
Eutrophic lakes are high in turbidity, nutrient inputs, and productivity while low in diversity and often anoxic. Evidence suggests the removal of periphyton from the zone of emergent vegetation is likely to push a lake into the eutrophic state. This evidence works under the assumption that vegetation provides significant nutrient uptake. Further research is needed to confirm that vegetation uses both nitrogen and phosphorus in sufficient proportions to limit their availability. If these nutrients are limited but not because of vegetative nutrient uptake, then this hypothesis will not hold up.
Periphyton in the zone of emergent vegetation uses CO2 from the atmosphere and nutrients from the lake system. If these plants are removed, nutrient availability in the lake may initially increase. Higher nutrient availability could benefit both macrophytes and algae populations. The significant difference between NPP in the N+P and control bottles provides clues about the existing nutrients in the lakes. Photosynthesis of autotrophs in the lakes can be derived from the net primary productivity. NPP was much higher in the bottles where both nitrogen and phosphorus were added. This indicates that autotrophic photosynthesis was likely limited by one or both nutrients. Figure 2 shows that NPP in the N+P was higher than either the N or P bottles. This supports the hypothesis that the lakes are co-limited by both nitrogen and phosphorus. Thus, removing the weeds as nutrient competitors may benefit other macrophytes populations in the lake. Algae are also nutrient-dependent and could benefit from less competition by periphyton for nutrients. If algae blooms are encouraged, the lake will become more turbid. Higher turbidity can prevent vegetation from reestablishing by limiting light availability in the littoral zones.
Increasing algae blooms, which increases turbidity, may also impact the temperature and DO profiles of the lakes. High turbidity results in low light penetration. If light is unable to reach below surface waters, the water underneath will not be as warm. This will result in overall colder, more stratified lakes with more extreme temperature profiles. If lakes are generally lower in temperature, there may be less evaporation of water from the lake surface. This could slow the rate of lake succession. If the algae populations increase, there will also be an increase of dying algal material that falls to the bottom of the lake. The detritivores living at the bottom of the lakes process the retired algae on the lake floor through respiration, converting food (dead algae) and DO into H2O and CO2. Increased dead algae at the bottom of the lake will increase decomposer populations and thus respiration. An increase in detritivore respiration will ultimately reduce the amount of DO, potentially resulting in an anoxic lake.
Macroinvertebrate populations may be most influenced by reductions in DO and vegetation. Lower levels of DO may ultimately inhibit macroinvertebrate populations since their respiration requires DO in the water. If other heterotrophs, such as detritivores at the lake bottom, outcompete macroinvertebrates for DO, the macroinvertebrate population may decrease. This would occur because less DO would limit macroinvertebrate respiration. It is likely that algae blooms will advantage detritivores and macroinvertebrates may die off from lack of DO in the lake. Vegetation may provide protection for macroinvertebrates from predation and disturbances. Periphyton can act as a physical barrier for macroinvertebrates, advantaging them in their predator-prey relationship with fish. This is only relevant in lakes that support fish that eat macroinvertebrates. Both the upper and lower lakes have been previously stocked with fish. All the fish in the upper lake are assumed to be dead, but the lower lake still contains some fish. Thus, this factor still may be relevant. The vegetation may also provide protection to macroinvertebrates from abiotic disturbances like wind.
Eutrophic lakes are high in turbidity, nutrient inputs, and productivity while low in diversity and often anoxic. Evidence suggests the removal of periphyton from the zone of emergent vegetation is likely to push a lake into the eutrophic state. This evidence works under the assumption that vegetation provides significant nutrient uptake. Further research is needed to confirm that vegetation uses both nitrogen and phosphorus in sufficient proportions to limit their availability. If these nutrients are limited but not because of vegetative nutrient uptake, then this hypothesis will not hold up.
Conclusion
Removal of vegetation from the littoral zone of shallow lakes may result in a shift from clear vegetated to eutrophic states. This management strategy could result in less competition for nutrients and thus higher nutrient concentrations. Increased nutrients might allow higher productivity for autotrophic populations including phytoplankton and algae. This may give algae an advantage which could lead to algae blooms and higher lake turbidity. Increases in turbidity could lower the water temperature and reduce evaporation, thus reducing the rate of succession. High algae populations may also result in increased rates of detritivore respiration and anoxic lakes. Finally, this could result in lower species diversity of macroinvertebrates. Removing periphyton may slow the rate of lake succession and simultaneously cause lakes to shift to the eutrophic state.
Works Cited
Chapin, F. S. et al. (2000). Consequences of changing biodiversity. Nature, 405(6783), 234-242.
Kochevar, R. (1987). The Catamount Ranch lakes: A comparative analysis of the aquatic habitat suitability. (BA thesis), Colorado College, Colorado Springs, CO.
Lewis, Simon L., and Mark A. Maslin. "Defining the anthropocene." Nature 519.7542 (2015): 171-180.
McCulloch, D. L. Introduction to Lentic Ecosystems. Environmental Biology class notes.
Biology department. Collin College. McKinney, TX.
Pelletier, M. C., Ebersole, J., Mulvaney, K., Rashleigh, B., Gutierrez, M. N., Chintala, M., ... & Lane, C. (2020). Resilience of aquatic systems: Review and management implications. Aquatic Sciences, 82(2), 1-25.
Penaluna, B. E., Olson, D. H., Flitcroft, R. L., Weber, M. A., Bellmore, J. R., Wondzell, S. M., ... & Reeves, G. H. (2017). Aquatic biodiversity in forests: a weak link in ecosystem services resilience. Biodiversity and Conservation, 26(13), 3125-3155.
Scheffer, M. (2009). Lakes. In Critical transitions in nature and society. (Vol. 16 pp. 109-138).
Princeton University Press.
Woolway, R. I., Kraemer, B. M., Lenters, J. D., Merchant, C. J., O’Reilly, C. M., & Sharma, S.
(2020). Global lake response to climate change. Nature Reviews Earth & Environment, 1(8), 388-403.
Zalasiewicz, J., Waters, C. N., Ellis, E. C., Head, M. J., Vidas, D., Steffen, W., Thomas, J. A., Horn, E., Summerhayes, C. P., Leinfelder, R., McNeill, J. R., Gałuszka, A., Williams, M., Barnosky, A. D., Richter, D. D. B., Gibbard, P. L., Syvitski, J., Jeandel, C., Cearreta, A., . . . Zinke, J. (2021). The anthropocene: Comparing its meaning in geology (chronostratigraphy) with conceptual approaches arising in other disciplines. Earth’s Future, 9(3). https://doi.org/10.1029/2020ef001896
Kochevar, R. (1987). The Catamount Ranch lakes: A comparative analysis of the aquatic habitat suitability. (BA thesis), Colorado College, Colorado Springs, CO.
Lewis, Simon L., and Mark A. Maslin. "Defining the anthropocene." Nature 519.7542 (2015): 171-180.
McCulloch, D. L. Introduction to Lentic Ecosystems. Environmental Biology class notes.
Biology department. Collin College. McKinney, TX.
Pelletier, M. C., Ebersole, J., Mulvaney, K., Rashleigh, B., Gutierrez, M. N., Chintala, M., ... & Lane, C. (2020). Resilience of aquatic systems: Review and management implications. Aquatic Sciences, 82(2), 1-25.
Penaluna, B. E., Olson, D. H., Flitcroft, R. L., Weber, M. A., Bellmore, J. R., Wondzell, S. M., ... & Reeves, G. H. (2017). Aquatic biodiversity in forests: a weak link in ecosystem services resilience. Biodiversity and Conservation, 26(13), 3125-3155.
Scheffer, M. (2009). Lakes. In Critical transitions in nature and society. (Vol. 16 pp. 109-138).
Princeton University Press.
Woolway, R. I., Kraemer, B. M., Lenters, J. D., Merchant, C. J., O’Reilly, C. M., & Sharma, S.
(2020). Global lake response to climate change. Nature Reviews Earth & Environment, 1(8), 388-403.
Zalasiewicz, J., Waters, C. N., Ellis, E. C., Head, M. J., Vidas, D., Steffen, W., Thomas, J. A., Horn, E., Summerhayes, C. P., Leinfelder, R., McNeill, J. R., Gałuszka, A., Williams, M., Barnosky, A. D., Richter, D. D. B., Gibbard, P. L., Syvitski, J., Jeandel, C., Cearreta, A., . . . Zinke, J. (2021). The anthropocene: Comparing its meaning in geology (chronostratigraphy) with conceptual approaches arising in other disciplines. Earth’s Future, 9(3). https://doi.org/10.1029/2020ef001896