- Home
- About Me
- Theme 1
- Theme 2
- Theme 3
-
Theme 4
-
Theme 5
- Application of Bloom’s Taxonomy in Outdoor Learning School
- Assessment Informing Instruction
- Developmental Pedagogy Assessment (LDPA) Question 5
- Reflections >
- An Evaluation of Systems Thinking in the 2021 TREE Semester Curriculum
- Recommendations for Equity-Focused Environmental Education Program Evaluations
ESSENTIAL QUESTION(S):
SUBJECT: Chemistry
GRADE LEVEL: 9th to 12th grade
CONCEPTS:
TIME ESTIMATE: 1 hour 15 minutes
LEARNING STYLE: Visual, auditory, hands-on
VOCABULARY:
LESSON SUMMARY:
This lesson focuses on introducing students to the process of following a procedure as part of a science experiment. The lesson begins with a short ice breaker before moving into the bulk of the lesson, performing a calorimetry lab. The class concludes with a guided calculation activity to test the accuracy of the experiment.
STUDENT LEARNING OBJECTIVES:
Students will be able to:
MATERIALS:
BACKGROUND INFORMATION:
The law of Conservation of Energy states that energy can neither be created nor destroyed. Energy cannot be lost, only converted from one form to another. Some common forms of energy are chemical, mechanical, potential, kinetic, radiant, and thermal. When energy is gained by a system, it was not created but transferred from one system to another. This is the First Law of Thermodynamics: heat energy loss by 1 body is gained by another.
Heat can be transferred by conduction, convection or radiation. When heat is transferred by conduction, the mediums are directly touching each other. One example would be heating up a metal spoon in hot soup. Convection describes heat transfer from the movement of a fluid such as water or air. One example would be heating the air in a hot air balloon causing it to fly because hot air is less dense than cold air and thus rises. Radiation is heat transferred through electromagnetic waves. One example would be feeling warm from the sun's rays.
Specific heat is the amount of energy needed by an object to raise the temperature by 1 degree Celsius. This is used to determine the calorie content of food samples. The specific heat of water is 1 cal/g.
When food burns, the stored energy (chemical energy) is converted to thermal energy and transferred to the water through convection. Thus, the temperature change of the water can be used to measure the amount of energy transferred from food. Energy transfer is calculated with equation 1.
Equation 1: Q=mCΔT
Q: heat
m: mass in grams
C: specific heat of water in cal/g
ΔT: change in water temperature, T (final)-T (initial)
COLORADO ACADEMIC STANDARDS ADDRESSED:
Standard 1: Physical Science-Academic Context and Connections.2
2. Plan and conduct an investigation individually and collaboratively to produce data to serve as the basis for evidence, and in the design: decide on types, how much, and accuracy of data needed to produce reliable measurements and consider limitations on the precision of the data. (e.g., number of trials, cost, risk, time), and refine the design accordingly (Planning and Carrying Out Investigations) (Personal: Personal responsibility)
Standard 1: Physical Science-Evidence Outcomes.
b. Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperature are combined within a closed system results in a more uniform energy distribution among the components in the system (second law of thermodynamics). (HS-PS3-4) (Clarification Statement: Emphasis is on analyzing data from student investigations and using mathematical thinking to describe the energy changes both quantitatively and conceptually. Examples of investigations could include mixing liquids at different initial temperatures or adding objects at different temperatures to water.) (Boundary Statement: Limited to investigations based on materials and tools provided to students.)
ASSESSMENT SUGGESTIONS:
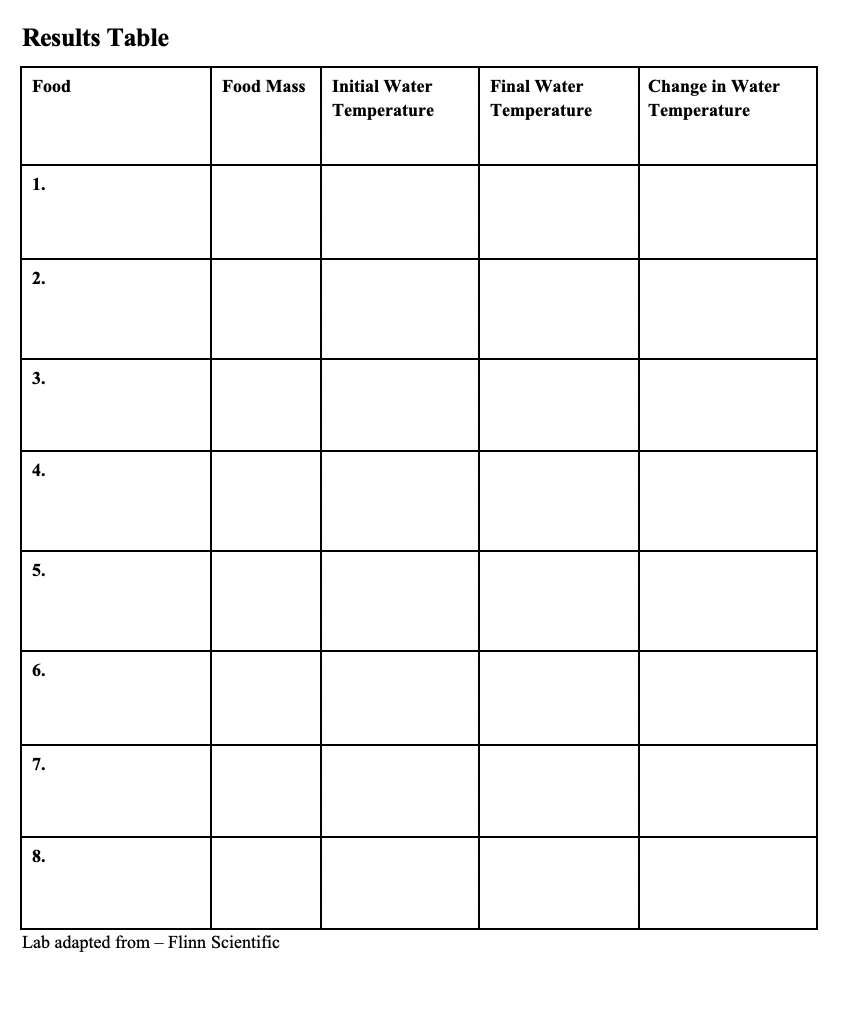
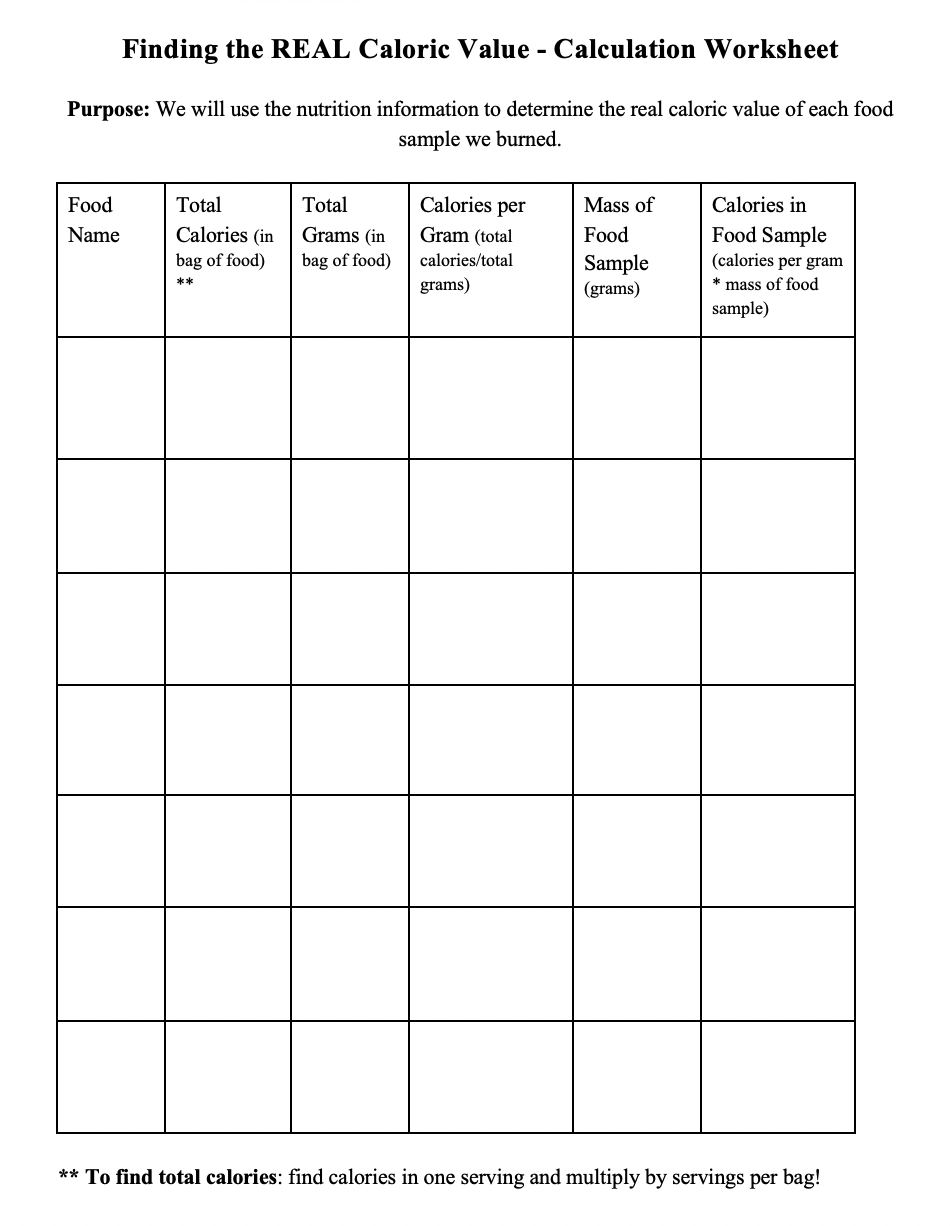
Students will complete the experiment in small groups and be able to verbally reflect on the experiment. Students will discuss whether they can improve the procedure and if so, what they will do to make the experiment more accurate. This discussion will happen in small groups. Each group will share a brief summary of their discussion with the class. At the end of the lesson, students will complete a post-experiment worksheet (see Appendix II).
ADVANCE PREPARATION:
Teachers should have lab procedures and materials ready before class. Teachers should test different foods to make sure they burn (Ex. Popcorn, Cheetos, fruit). Teachers should use the worksheet to perform practice calculations to make sure it is correct.
RESOURCES/REFERENCES:
Flinn Scientific Inc. (2016). Soda can calorimeter. https://www.flinnsci.com/api/library/Download/f9560a5fc7ef4a6b8f4598fea30626eb
LITERATURE:
Santone, S. (2018). Reframing the curriculum: Design for social justice and sustainability (kappa delta pi Co-Publications) (1st ed.). Routledge.
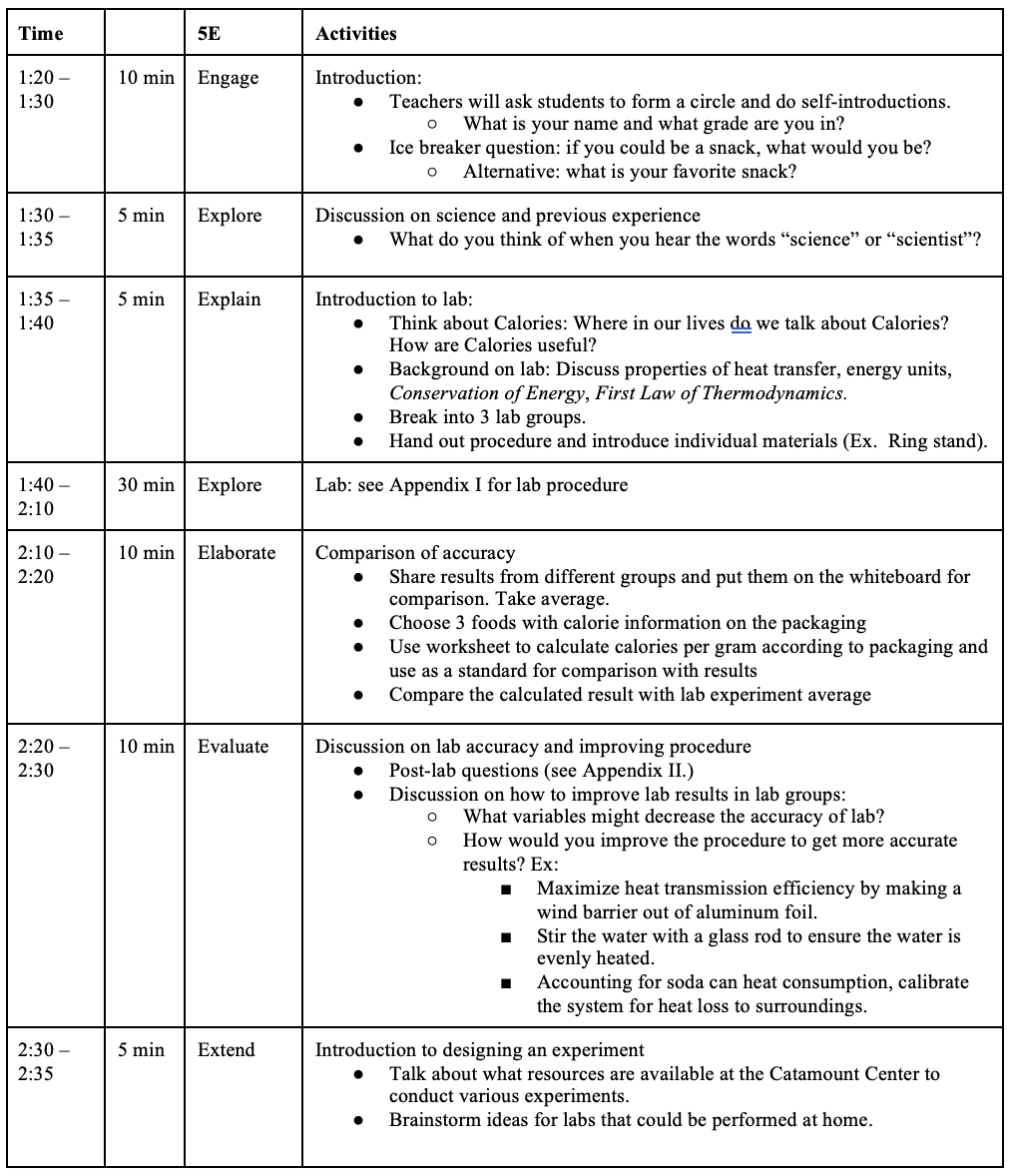
PROCEDURE AND DISCUSSION QUESTIONS WITH TIME ESTIMATE:
- What is the scientific process?
- How can a scientific experiment be conducted?
SUBJECT: Chemistry
GRADE LEVEL: 9th to 12th grade
CONCEPTS:
- Following a scientific procedure
- Using Scientific Experiments to explore real-world problems.
- Community relationships
TIME ESTIMATE: 1 hour 15 minutes
LEARNING STYLE: Visual, auditory, hands-on
VOCABULARY:
- Experiment: A procedure that tests a hypothesis.
- Procedure: Steps you follow towards an outcome.
- Hypothesis: A proposed explanation based on information, often in the format of “if… then… because….”
- Calorimetry: The process of measuring heat transfer in and out of a system.
- calorie: often written as cal, it is the amount of energy required to raise 1 g of water by 1-degree Celsius.
- Calories: often written as kcal, it equals 1000 cal. Often used in nutrition facts.
- Heat capacity: the amount of heat required to raise the temperature of an object by 1 degree Celsius. The specific heat capacity for water is 1cal/g.
LESSON SUMMARY:
This lesson focuses on introducing students to the process of following a procedure as part of a science experiment. The lesson begins with a short ice breaker before moving into the bulk of the lesson, performing a calorimetry lab. The class concludes with a guided calculation activity to test the accuracy of the experiment.
STUDENT LEARNING OBJECTIVES:
Students will be able to:
- Follow a procedure.
- Reflect on an experiment and improve the procedure.
- Calculate the calories of individual food using the materials provided for the calorimetry lab.
- Identify individuals or groups that may benefit from learning about the results of the experiment
MATERIALS:
- 3 ring stands
- 3 soda cans
- 3 corks
- 3 needles
- 3 weighing balances
- 3 thermometers
- 3 lighters
- 1 Erlenmeyer Flask
- 1 coffee can
- 1 roll of tin foil
- Variety of food: rice, Cheeto, chip, popcorn, dried fruit, spaghetti, bacon, crouton…
- 3 50 mL graduated cylinder
- 15 procedure sheets
- 15 calculation sheet
- 15 safety goggles
BACKGROUND INFORMATION:
The law of Conservation of Energy states that energy can neither be created nor destroyed. Energy cannot be lost, only converted from one form to another. Some common forms of energy are chemical, mechanical, potential, kinetic, radiant, and thermal. When energy is gained by a system, it was not created but transferred from one system to another. This is the First Law of Thermodynamics: heat energy loss by 1 body is gained by another.
Heat can be transferred by conduction, convection or radiation. When heat is transferred by conduction, the mediums are directly touching each other. One example would be heating up a metal spoon in hot soup. Convection describes heat transfer from the movement of a fluid such as water or air. One example would be heating the air in a hot air balloon causing it to fly because hot air is less dense than cold air and thus rises. Radiation is heat transferred through electromagnetic waves. One example would be feeling warm from the sun's rays.
Specific heat is the amount of energy needed by an object to raise the temperature by 1 degree Celsius. This is used to determine the calorie content of food samples. The specific heat of water is 1 cal/g.
When food burns, the stored energy (chemical energy) is converted to thermal energy and transferred to the water through convection. Thus, the temperature change of the water can be used to measure the amount of energy transferred from food. Energy transfer is calculated with equation 1.
Equation 1: Q=mCΔT
Q: heat
m: mass in grams
C: specific heat of water in cal/g
ΔT: change in water temperature, T (final)-T (initial)
COLORADO ACADEMIC STANDARDS ADDRESSED:
Standard 1: Physical Science-Academic Context and Connections.2
2. Plan and conduct an investigation individually and collaboratively to produce data to serve as the basis for evidence, and in the design: decide on types, how much, and accuracy of data needed to produce reliable measurements and consider limitations on the precision of the data. (e.g., number of trials, cost, risk, time), and refine the design accordingly (Planning and Carrying Out Investigations) (Personal: Personal responsibility)
Standard 1: Physical Science-Evidence Outcomes.
b. Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperature are combined within a closed system results in a more uniform energy distribution among the components in the system (second law of thermodynamics). (HS-PS3-4) (Clarification Statement: Emphasis is on analyzing data from student investigations and using mathematical thinking to describe the energy changes both quantitatively and conceptually. Examples of investigations could include mixing liquids at different initial temperatures or adding objects at different temperatures to water.) (Boundary Statement: Limited to investigations based on materials and tools provided to students.)
ASSESSMENT SUGGESTIONS:
Students will complete the experiment in small groups and be able to verbally reflect on the experiment. Students will discuss whether they can improve the procedure and if so, what they will do to make the experiment more accurate. This discussion will happen in small groups. Each group will share a brief summary of their discussion with the class. At the end of the lesson, students will complete a post-experiment worksheet (see Appendix II).
ADVANCE PREPARATION:
Teachers should have lab procedures and materials ready before class. Teachers should test different foods to make sure they burn (Ex. Popcorn, Cheetos, fruit). Teachers should use the worksheet to perform practice calculations to make sure it is correct.
RESOURCES/REFERENCES:
Flinn Scientific Inc. (2016). Soda can calorimeter. https://www.flinnsci.com/api/library/Download/f9560a5fc7ef4a6b8f4598fea30626eb
LITERATURE:
Santone, S. (2018). Reframing the curriculum: Design for social justice and sustainability (kappa delta pi Co-Publications) (1st ed.). Routledge.
PROCEDURE AND DISCUSSION QUESTIONS WITH TIME ESTIMATE: